Zoll - LifeVest Wearable Defib
by Zoll
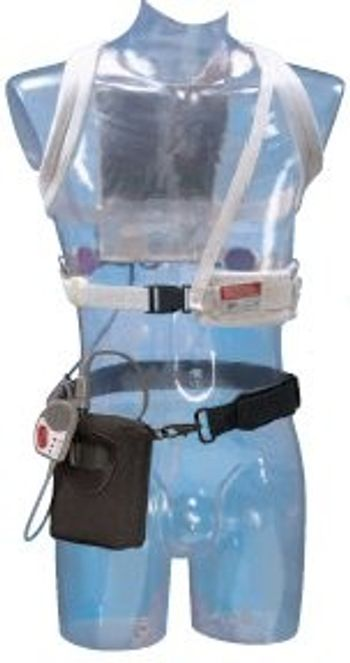
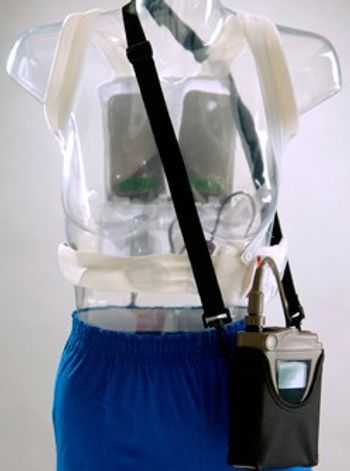


The LifeVest (Life Vest) wearable defibrillator is the first and only wearable defibrillator for patients at high risk of sudden cardiac arrest (SCA). The non-invasive device, approved by the FDA in 2002, provides emergency defibrillation and has been worn by over 9,000 patients, and has a 98 percent first shock success rate for treating patients for SCA without requiring bystander intervention. Timely emergency defibrillation is the single most important factor in saving a SCA victim's life. If it detects a life threatening rhythm, the LifeVest alerts the patient to allow a conscious patient to prevent a shock. If the patient is unconscious, the device delivers emergency defibrillation in the form of an electrical shock to restore normal rhythm. The entire event, from detecting a life-threatening arrhythmia to automatically delivering an emergency defibrillation shock, usually occurs in less than a minute. [b]Life Vest Emergency Defibrillation [/b] The LifeVest wearable defibrillator is worn outside the body rather than implanted in the chest. The LifeVest system consists of two main components: (1) an electrode belt and garment that surrounds the patient's chest, and (2) a monitor that the patient wears around the waist or from a shoulder strap. The LifeVest electrodes are dry and non-adhesive to provide patient comfort. The monitor weighs about 1.8 pounds, making it the lightest external emergency defibrillator available.
2Replies2 years ago | Manual I need a maualReply |
0Replies8 years ago | Application of vest when inpatient Should a life vest be worn when the patient is hospitalized on a telemetry unit ? Reply |
0Replies9 years ago | Wearable defibrillator design project
Hi all!
Liam Reply |
[list] [*] Constant Monitoring - Records critical patient data to help diagnose and treat arrhythmic conditions [*] Provides continuous ECG and patient compliance monitoring [*] Immediate Protection - Advanced VT/VF detection algorithm tailored to the patient [*] Fully-automated system that offers less-than-one-minute treatment for unresponsive patients suffering from a tachyarrhythmia [*] Peace of Mind - Protects your patient while determining the need for an ICD [*] Fully automatic - doesn't require bystander response or intervention from anyone [/list]
| Defibrillator Type | External, Manual |
In January 2018, patients and health care providers were informed of a potential problem in the charging circuitry of the LifeVest 4000, which may prevent it from delivering a life-saving shock to patients wearing the device.