Medtronic - MindFrame Capture
by Medtronic
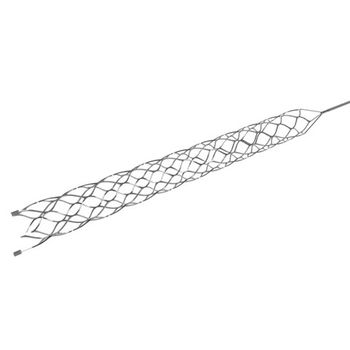

The MindFrame Capture™ LP device is a stent retriever thrombectomy device designed to navigate, access, and treat distal zone occlusions in patients.
The MindFrame Capture LP revascularization device is intended to restore blood flow by removing thrombus from a large intracranial vessel in patients experiencing ischemic stroke within 8 hours of symptom onset. Patients who are ineligible for intravenous tissue plasminogen activator (IV t-PA) or who fail IV t-PA therapy are candidates for treatment. This device should only be used by physicians trained in interventional neuroradiology and treatment of ischemic stroke.
The MindFrame Capture LP revascularization device features distal and proximal markers for accurate positioning and a proprietary cell geometry that minimizes deformation and maximizes clot entrapment.1 The only stent retriever thrombectomy system compatible with a catheter in the 10/14 class.
The FDA has identified this as a Class I recall, the most serious type of recall. Use of these devices may cause serious injuries or death.