CORPAK Medsystems - CORFLO Anti IV Enteral Feeding System
by CORPAK Medsystems
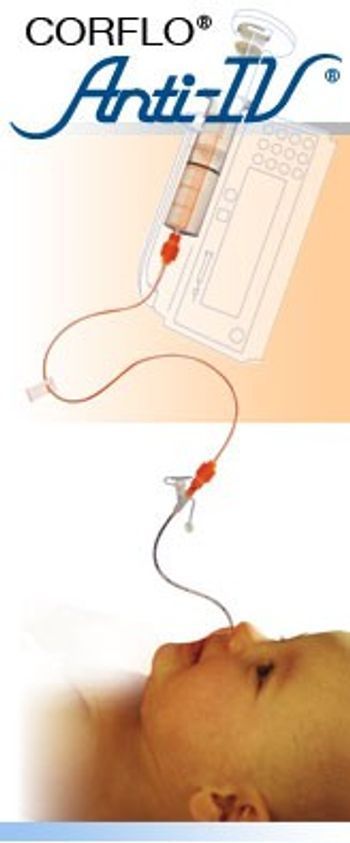
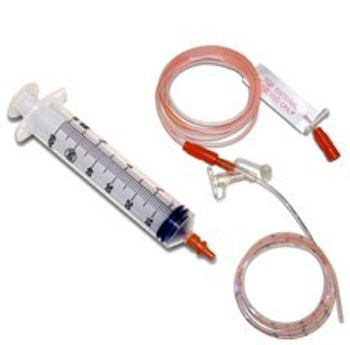


The CORFLO® Anti-IV Feeding Tube and Extension Set can be used with any feeding method. The proprietary connection on the feeding tube will allow feeding only through the CORFLO Anti-IV Feeding Set to help ensure Enteral Only Use. The FARRELL Valve with Anti-IV Connectors is designed to help patients who suffer from poor gastric motility, pain and bloating. It connects to the CORFLO Anti-IV Enteral System.
0Replies13 years ago | Device Alert See Attached PDF under Manuals & Resources tab The Joint Commission Journal on Quality and Patient Safety Enteral Feeding Misconnections: A Consortium Position Statement USP Medication SafetyReply |
0Replies13 years ago | Device Alert See Attached PDF under Manuals & Resources tab Issue 36 - April 3, 2006 Tubing misconnections—a persistent and potentially deadly occurrence Tubing and catheter misconnection errors are an important and under-reported problem in health care organizations. In addition, these errors are often caught and corrected before any injury to the patient occurs. Given the reality of and potential for life threatening consequences, increased awareness and analysis of these errors—including averted errors—can lead to dramatic improvement in patient safety. To date, nine cases involving tubing misconnections have been reported to the Joint Commission’s Sentinel Event Database. These resulted in eight deaths and one instance of permanent loss of function, and affected seven adults and two infants. Reports in the media and to organizations such as ECRI, the Food and Drug Administration (FDA), the Institute for Safe Medication Practices (ISMP), and United States Pharmacopeia (USP) indicate that misconnection errors occur with significant frequency and, in a number of instances, lead to deadly consequences. Types of misconnections The types of tubes and catheters involved in the cases reported to the Joint Commission included central intravenous catheters, peripheral intravenous catheters, nasogastric feeding tubes, percutaneous enteric feeding tubes, peritoneal dialysis catheters, tracheostomy cuff inflation tubes, and automatic blood pressure cuff insufflation tubes. The specific misconnections involved an enteric tube feeding into an intravenous catheter (4 cases); injection of barium sulfate (GI contrast medium) into a central venous catheter (1 case); an enteric tube feeding into a peritoneal dialysis catheter (1 case); a blood pressure insufflator tube connected to an intravenous catheter (2 cases); and injection of intravenous fluid into a tracheostomy cuff inflation tube (1 case). A review by USP of more than 300 cases reported to its databases found misconnection errors involving the following: • Intravenous infusions connected to epidural lines, and epidural solutions (intended for epidural administration) connected to peripheral or central IV catheters. • Bladder irrigation solutions using primary intravenous tubing connected as secondary infusions to peripheral or central IV catheters. • Infusions intended for IV administration connected to an indwelling bladder (foley) catheter. • Infusions intended for IV administration connected to nasogastric (NG) tubes. • Intravenous solutions administered with blood administration sets, and blood products transfused with primary intravenous tubing. • Primary intravenous solutions administered through various other functionally dissimilar catheters, such as external dialysis catheters, a ventriculostomy drain, an amnio-infusion catheter, and the distal port of a pulmonary artery catheter. Many of the misconnection cases involved luer connectors—small devices used in the connection of many medical components and accessories. There are two types of luer connectors—slips and locks. A luer slip connector consist of a tapered “male” fitting that slips into a wider “female” fitting to create a secure connection. The luer lock connector has a threaded collar on the “male” fitting and a flange on the “female” fitting that screw together to create a more secure connection. Examples of misconnections involving luer connectors include the following: • Capnography sampling tube to an intravenous cannula. • Enteral feeding set to a central venous catheter. • Enteral feeding set to a hemodialysis line. • Noninvasive blood pressure (NIBP) insufflation tube to a needleless IV port. • Oxygen tubing to a needleless IV port. • Sequential compression device (SCD) hose to needleless “piggy-back” port of an IV administration set. Root causes identified The basic lesson from these cases is that if it can happen, it will happen. Luer connectors are implicated in or contribute to many of these errors because they enable functionally dissimilar tubes or catheters to be connected. Other identified causes include the routine use of tubes or catheters for unintended purposes, such as using IV extension tubing for epidurals, irrigation, drains, and central lines, or to extend enteric feeding tubes; and the positioning of functionally dissimilar tubes used in patient care in close proximity to one another. In the cases reported to the Sentinel Event Database, contributing factors included movement of the patient from one setting or service to another, and staff fatigue associated with working consecutive shifts. Risk reduction strategies There are currently no published standards that specifically restrict the use of luer connectors to certain medical devices. Consequently, a broad range of medical devices, which have different functions and access the body through different routes, are often outfitted with luer fittings that can be easily misconnected. Organizations in Europe and the U.S. are now developing standards to restrict the types of devices that use luer fittings in an attempt to mitigate misconnection hazards. According to Jim Keller, vice president, Health Technology Evaluation and Safety for ECRI, and Stephanie Joseph, project engineer for ECRI, the solution to reducing—even eliminating—misconnection errors lies in both engineering controls respecting how products and devices are designed (“incompatibility by design”), and in re-engineering work practices. “A well-designed device should prevent misconnections and should prompt the user to take the correct action,” explains Joseph, author of a guidance article published in the March 2006 issue of ECRI’s Health Devices journal. As a first step in prevention, Joseph urges hospitals to avoid buying non-intravenous equipment (such as nebulizers, NIBP devices, and enteral feeding sets) that can mate with the luer connectors on patient IV lines. In addition, Joseph emphasizes that the single most important work practice solution for clinicians is to trace all lines back to their origin before connecting or disconnecting any devices or infusions. Other solutions include specific education and training regarding this problem for all clinicians and having practitioners take simple precautions such as turning on the light in a darkened room before connecting or reconnecting tubes or devices. The risk of waking a sleeping patient is minimal by comparison. Errors have also occurred when patients or family members attempt to disconnect and reconnect equipment themselves. Staff should emphasize to all patients the importance of contacting a clinical staff member for assistance when there is an identified need to disconnect or reconnect devices. Other approaches to reducing the risk of misconnections that have been identified also have significant potential for unintended consequences. These include: • Labeling all tubes and catheters—This may not always be practical and may therefore lead to inconsistent implementation. However, the labeling of certain high-risk catheters (epidural, intrathecal, arterial) should always be done. • Color-coding tubes and catheters—This can lead users to rely on the color coding rather than assuring a clear understanding of which tubes and catheters are connected correctly to which body inlets. In addition, the training and educating of all staff (including temporary agency and travel staff) about the institution’s color-coding system requires continuing attention. Finally, color-coding schemes often vary across institutions in the same community, creating increased risk when agency and travel staff are used. Joint Commission recommendations The Joint Commission offers the following recommendations and strategies to health care organizations to reduce tubing misconnection errors: 1. Do not purchase non-intravenous equipment that is equipped with connectors that can physically mate with a female luer IV line connector. 2. Conduct acceptance testing (for performance, safety and usability) and, as appropriate, risk assessment (e.g., failure mode and effect analysis) on new tubing and catheter purchases to identify the potential for misconnections and take appropriate preventive measures. 3. Always trace a tube or catheter from the patient to the point of origin before connecting any new device or infusion. 4. Recheck connections and trace all patient tubes and catheters to their sources upon the patient’s arrival to a new setting or service as part of the hand-off process. Standardize this “line reconciliation” process. 5. Route tubes and catheters having different purposes in different, standardized directions (e.g., IV lines routed toward the head; enteric lines toward the feet). This is especially important in the care of neonates. 6. Inform non-clinical staff, patients and their families that they must get help from clinical staff whenever there is a real or perceived need to connect or disconnect devices or infusions. 7. For certain high-risk catheters (e.g., epidural, intrathecal, arterial), label the catheter and do not use catheters that have injection ports. 8. Never use a standard luer syringe for oral medications or enteric feedings. 9. Emphasize the risk of tubing misconnections in orientation and training curricula. 10. Identify and manage conditions and practices that may contribute to health care worker fatigue, and take appropriate action. In addition, the Joint Commission urges product manufacturers to implement “designed incompatibility,” as appropriate, to prevent dangerous misconnections of tubes and catheters. Resources ISMP Medication Safety Alert, June 17, 2004, www.ismp.org/MSAarticles/tubingprint.htm Nursing 2005, 35 (9), September 2005, pg. 73, by Melissa Eakle, R.N., MBA, MSN; Beverly Albrecht Gallauresi, R.N., B.S., MPH; and Audrey Morrison, R.N. FDA Patient Safety News, Show #31, September 2004; Show #20, October 2003; Show #46, December 2005 “Fatal Air Embolism Caused by the Misconnection of a Medical Device Hoses to Needleless Luer Ports on IV Administration Sets” [hazard report], ECRI, Health Devices, June 2004; 33(6):223-5 “Misconnected Flowmeter Leads to Two Deaths” [special report], ECRI, Health Devices Alerts, January 25, 2003 “Preventing Misconnections of Lines and Cables,” ECRI, Health Devices, March 2006; 35(3):81-95Reply |
[list] [*] Be compatible only with oral tip syringes [*] Prevent inadvertent feeding into an IV line [*] Prevent injecting IV medication into the feeding port of the feeding tube [*] Prevent direct connection of the feeding set or NG tube to any standard Luer lock or Luer slip syringe or connector [/list]
See Attached PDF under Manuals & Resources tab The Joint Commission Journal on Quality and Patient Safety Enteral Feeding Misconnections: A Consortium Position Statement USP Medication Safety
See Attached PDF under Manuals & Resources tab Issue 36 - April 3, 2006 Tubing misconnections—a persistent and potentially deadly occurrence Tubing and catheter misconnection errors are an important and under-reported problem in health care organizations. In addition, these errors are often caught and corrected before any injury to the patient...