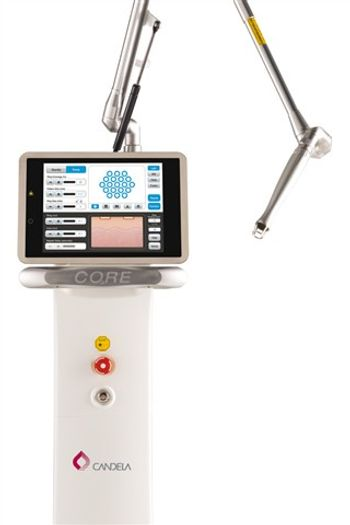
Syneron-Candela - CO2RE
Syneron-Candela CO2RE Forums
Add Aesthetic Laser to My Bench5 Results
Updated Apr 4 2024
2 Replies
After using the laser for about 10 minutes, the screen slowly stops responding to touch commands. Is this due to the touch screen itself failing, or is there a board that needs to be replaced? Anyone know the generic service codes for the CO2re? It has the old software. harrisbw@hotmail.com
RE: Syneron-Candela - CO2REUpdated Aug 8 2022
4 RepliesTrying to repair my co2re. The aiming beam is not centered properly. I've got the thing opened up. Any advice on aligning the aiming beam properly? I'm having a tough time...
Josh@thelaseragent.com
Equipment: Syneron-Candela - CO2RE
RE: Syneron-Candela - CO2REAdded May 6 2017
Added Dec 4 2015
0 Replies
Does anyone have a manual either operational or service. Or can someone tell me how to and how often we should do safety checks on this equipment.
RE: Syneron-Candela - CO2REAdded Jan 4 2011
0 RepliesNew fractional CO2 resurfacing laser offers practitioners broader and highly effective treatment options
YOKNEAM, Israel and IRVINE, Calif., Nov. 30, 2010 /PRNewswire/ -- Syneron Medical Ltd. (Nasdaq: ELOS), www.syneron.com, the global leader in the medical aesthetic device marketplace, announced today the US Food and Drug Administration (FDA) and Health Canada clearance and commercial launch of its highly flexible CO2RE CO2 resurfacing system. CO2RE is a versatile fractional CO2 system that offers the unique ability to treat both superficial and deep skin layers simultaneously with precision-control over the intensity, pattern and depth of ablation. This patent-pending technology is the first to receive FDA clearance for all of its operating modes, including the four major fractional treatment modes. The system can be used for fractional CO2 resurfacing and traditional ablation, and is ideal for skin rejuvenation, wrinkle reduction, scar reduction and other aesthetic procedures.
“We received very positive physician feedback during the international roll-out of the CO2RE system and we are excited to begin offering it in the United States and Canada, giving our customers more choices and greater flexibility to meet their individual practice needs. With the addition of CO2RE, we now offer a complete range of product solutions that address physicians’ and patients' needs across a wide range of facial rejuvenation treatment categories,” said Louis P. Scafuri, Chief Executive Officer of Syneron. Mr. Scafuri added, “The launch of CO2RE shows the strength of our globalized approach to research and development. The product was developed by Inlight Inc., San-Diego, Calif. which was acquired by Syneron at the end of 2008. Inlight brought to Syneron the most advanced scanner and ablative laser technology which, in combination with Syneron’s know-how in light tissue interaction, enabled us to bring to the market the most advanced and versatile system for a wide range of ablative skin rejuvenation procedures.”
"We've been very happy with the results that we've seen using CO2RE. It has a lot of versatility and we find it works well for both fractionated ablative resurfacing as well as traditional ablative resurfacing," said Brian Zelickson, MD, a board certified dermatologist at the Zel Skin and Laser Specialists.
E. Victor Ross, MD, at the Scripps Clinic Carmel Valley Laser and Cosmetic Dermatology commented, "One of CO2RE's advantages is the handpiece, which is very easy to hold. In comparison to other fractional handpieces the CO2RE handpiece is thin, lightweight and easy to maneuver around the skin during treatments."
About CO2RE
The new CO2RE fractional CO2 resurfacing system is a lighter, faster and more versatile solution for skin resurfacing, wrinkle and scar reduction, and other traditional CO2 applications. CO2RE is the only CO2 laser to offer multi-depth pulse technology, delivering precisely fractionated beam patterns to treat multiple layers of the epidermis and dermis simultaneously.
About Syneron Medical Ltd.
Syneron Medical Ltd. – a company devoted to real technology, real science and real results – is the leading global aesthetic device company with a comprehensive product portfolio and a global distribution footprint. The Company's technology enables physicians to provide advanced solutions for a broad range of medical-aesthetic applications including body contouring, hair removal, wrinkle reduction, rejuvenation of the skin's appearance through the treatment of superficial benign vascular and pigmented lesions, and the treatment of acne, leg veins and cellulite. The Company sells its products under two distinct brands, Syneron and Candela. With its innovative approach to aesthetic treatments, Syneron has now entered into one of the largest in-demand applications, skin lightening. Founded in 2000, the corporate, R&D and manufacturing headquarters for Syneron Medical Ltd. are located in Israel. Syneron also has R&D and manufacturing operations in the US. The company markets and services and supports its products in 86 countries. It has offices in North America, France, Germany, Italy, Portugal, Spain, UK, Australia, China, Japan, and Hong Kong and distributors worldwide.
Additional information can be found at www.syneron.com.
[url=http://www.prnewswire.com/news-releases/syneron-announces-us-and-canada-launch-of-co2re-system-following-fda-and-health-canada-clearance-for-the-system-111019629.html]Article Source [/url] RE: Syneron-Candela - CO2RE